To print view
2016年11月24日更新
About The Fukushima Medical Device Development Support Centre

The Fukushima Medical Device Development Support Centre is established by Fukushima Prefecture as the first hub centre in Japan for integrally supporting implementation of the process starting from development to commercialisation of medical devices.
The Centre pursues the recovery of Fukushima Prefecture through the promotion of the industries related to medical devices and contribute to the development of the medical device industry in the world.
The Centre pursues the recovery of Fukushima Prefecture through the promotion of the industries related to medical devices and contribute to the development of the medical device industry in the world.
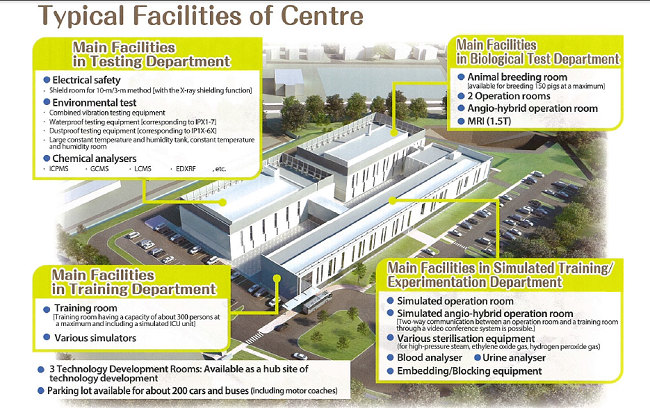
Functions
Safety Assessment Function

[Biological safety test]
Availablefor implantation test using animals (e.g. pigs) in accordance with the guidelines of the Japan Ministry of Health, Labour and Welfare.
[Non-biologicalsafety test]
Available for electrical safety test based on IEC60601, a variety of environmental tests, analysis of substances under RoHS (Restriction on Hazardous Substances).
Availablefor implantation test using animals (e.g. pigs) in accordance with the guidelines of the Japan Ministry of Health, Labour and Welfare.
[Non-biologicalsafety test]
Available for electrical safety test based on IEC60601, a variety of environmental tests, analysis of substances under RoHS (Restriction on Hazardous Substances).
Human Resource Development/ Traning Function
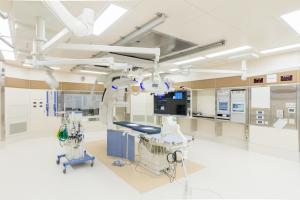
To support procedural training for physicians using pigs and humane body models and education/ training for health-care workers in operation rooms with angiography and simulated ICU unit.
* Equipment for washing, disinfection, sterilisation is available for the trainings.
To provide places for organising seminars and training for "manufacturing companies" inside or outside of Fukushima Prefecture that are planning to enter the field of medical devices or start overseas operations.
* Equipment for washing, disinfection, sterilisation is available for the trainings.
To provide places for organising seminars and training for "manufacturing companies" inside or outside of Fukushima Prefecture that are planning to enter the field of medical devices or start overseas operations.
Consulting/ Information Transmission Functions
To providelegal advice/ guidance on related laws and regulations such as the “Law for Ensuring the Quality, Efficacy, and Safety of Drugs and Medical Devices" for small and medium-sized companies that are planning to start the development or commercialisation of medical devices by experts.
Maching Function

To support matching between companies for supplying components to medical device manufacturers, coordinating mass-production/ OEM supply, organising exhibitions.
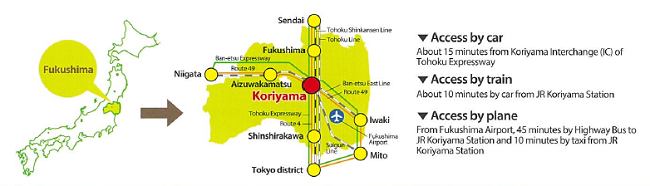
Location:
Contacts:
Tel: (+81) 24-954-4011